Natural Photosynthesis
Photosynthesis is a chemical process that occurs in photoautotrophs (organisms that make their own food), in which light energy is converted into sugars and other organic compounds. It consists of a series of chemical reactions that require carbon dioxide and water to begin. The light energy that hits the photoautotrophs is absorbed, and drives these chemical reactions to produce carbohydrates and oxygen as a by-product. The following equation is the basis of photosynthesis:
6CO2 + 6H2O + light energy ----> C6H12O6 + 6O2
6CO2 + 6H2O + light energy ----> C6H12O6 + 6O2
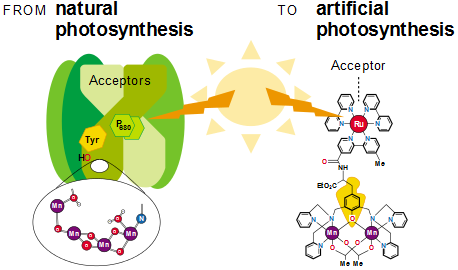
In natural photosynthesis, P680 absorbs photons of light, however, in many attempts at artificial photosynthesis, scientists have seen that the metal Ruthenium can replace P680 as the light absorber. In both takes on photosynthesis, it is seen that a Manganese complex sends electrons back to the light absorbing molecules, creating a surface for water-splitting
Artificial photosynthesis
Scientists across the world have been trying to replicate the natural reactions that occur during photosynthesis, and have come across the science of artificial photosynthesis. The term artificial photosynthesis is used to refer to any mechanism made to capture light and store energy from the Sun in chemical bonds of a solar fuel
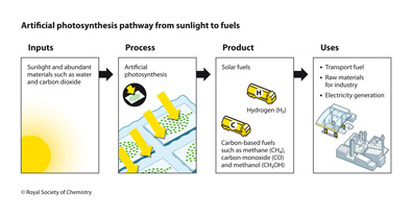
Artificial Photosynthesis Pathways: From sunlight to fuel
1.) The first step in artificial photosynthesis is for the reactants come together. The reactants include sunlight along with water and carbon dioxide that is available in the atmosphere
2.) These reactants then go through the whole process of photosynthesis artificially. Scientists have been known to use artificial leaves to split water, producing both oxygen and hydrogen. They are now creating artificial leaves using ruthenium and manganese complexes to mimic the natural process of photosynthesis (electron transport, etc.)
3.) The products of this reaction are created as soon as water is split, producing both oxygen and hydrogen. The hydrogen is then either used directly as a fuel or is used to reduce carbon dioxide and produce organic fuels
2.) These reactants then go through the whole process of photosynthesis artificially. Scientists have been known to use artificial leaves to split water, producing both oxygen and hydrogen. They are now creating artificial leaves using ruthenium and manganese complexes to mimic the natural process of photosynthesis (electron transport, etc.)
3.) The products of this reaction are created as soon as water is split, producing both oxygen and hydrogen. The hydrogen is then either used directly as a fuel or is used to reduce carbon dioxide and produce organic fuels